Phase transitions in nanostructured water confined in carbon nanotubes by external electric and magnetic fields: a molecular dynamics investigation†
Abstract
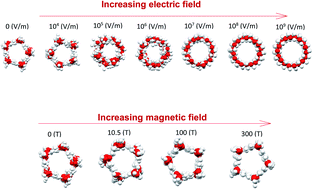
Applying electric and magnetic fields on water molecules confined in carbon nanotubes (CNTs) has important applications in cell biology and nanotechnology-based fields. In this work, molecular dynamics (MD) simulations were carried out to examine the probable phase transitions in confined water molecules confined in (14,0) CNTs at 300 K by applying different electric and magnetic fields in the axial direction. We have also studied some thermodynamics and structural properties of the confined water molecules in the different fields. Our results showed that the confined water molecules experience. Some phase (shape) transitions from the pentagonal to twisted pentagonal, spiral and circle-like shapes by increasing the electric field from 104 (V m−1) to 107 (V m−1). Also, applying the magnetic field with different intensities has small effects on the pentagonal shape of confined water molecules but applying the highest magnetic field (300 T) makes the pentagonal shape more ordered. These phase transitions have not been reported before. Our results also indicated that the ring-like shapes obtained in the presence of the electric field form more hydrogen bonds (HBs) than the other structures. The phase transitions of confined water molecules have been also proved by radial distribution function (RDF) and angle distribution function (ADF) analyses.


 Please wait while we load your content...
Please wait while we load your content...