Perylene bisimide cyclophanes as receptors for planar transition structures – catalysis of stereoinversions by shape-complementarity and noncovalent π–π interactions†
Abstract
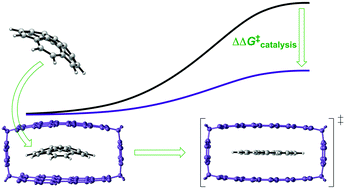
Perylene bisimide (PBI) cyclophanes have been shown to form ideal hosts for large aromatic molecules, offering rigid cavities and ideal monomer spacing for π–π stacking interactions between the host and guest. Here, we show computationally that these characteristics also make PBI cyclophanes ideal receptors for the planar transition structures of a number of representative stereoinversion processes. The observed catalytic effect can hereby be explained using two fundamental concepts: transition state stabilization due to shape-complementarity between cyclophane and the transition structures and, to a smaller extent, ground state destabilization due to non-complementarity between cyclophane and equilibrium structures. Energy decomposition analyses of the noncovalent interactions between the host and guest structures reveal that dispersion and electrostatic interactions make the largest contributions to stabilizations and thus suggest that catalysis in this system is mainly driven by π–π stacking interactions.
- This article is part of the themed collection: Macrocycle-based Supramolecular Elements


 Please wait while we load your content...
Please wait while we load your content...