Recent advances in transition metal-catalyzed reactions of carbamoyl chlorides
Abstract
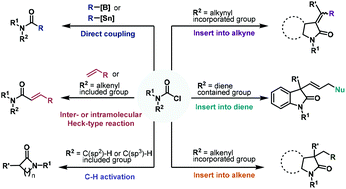
The development of carbamoyl chlorides as synthons for various amide-containing molecules and heterocycles has been summarized and discussed in this review article. Carbamoyl chlorides can participate in a diverse range of transition metal-catalyzed transformations including radical initiated reactions and cross-coupling and annulation reaction modes as well as C–H functionalization to access amide-functionalized organic frameworks having extensive applications in pharmaceutical and natural product synthesis. This review mainly highlights the advancements of carbamoyl chlorides as a powerful synthetic tool in transition-metal catalyzed reactions over the last two decades. In addition, transition-metal catalyzed reactions of their corresponding structural analogs, carbamoyl fluorides and cyanides, with respect to their chemical behavior are also briefly reviewed.


 Please wait while we load your content...
Please wait while we load your content...