Cascade intramolecular rearrangement/cycloaddition of nitrocyclopropane carboxylates with alkynes/alkenes: access to uncommon bi(hetero)cyclic systems†
Abstract
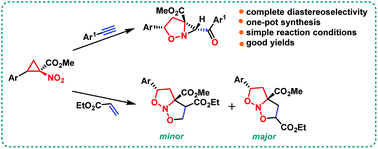
A straightforward approach for the one-pot synthesis of aziridinoisoxazoles via cascade intramolecular rearrangement of nitrocyclopropane carboxylates to cyclic nitronates followed by a thermal cycloaddition reaction with substituted phenylacetylenes has been demonstrated. All of the synthesized aziridinoisoxazoles were mostly obtained as single diastereomers, indicating a highly diastereoselective nature of the protocol. Additionally, nitrocyclopropane carboxylates were also made to react with ethyl acrylate to synthesize isoxazolo[2,3-b]isoxazole derivatives in a cascade one-pot fashion. The reactivity of nitrocyclopropane carboxylates with acetylenes and acrylates showed the synthetic divergence of the protocol towards the efficient synthesis of a less explored class of nitrogen heterocycles.


 Please wait while we load your content...
Please wait while we load your content...