Design of (β-diazo-α,α-difluoroethyl)phosphonates and their application as masked carbenes in visible light-promoted coupling reactions with sulfonic acids†
Abstract
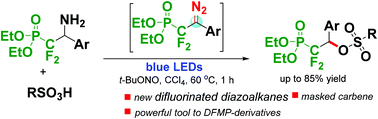
We elaborate a method for the generation of (2-diazo-1,1-difluoroethyl)phosphonates from (β-amino-α,α-difluoroethyl)phosphonates as precursors and their application as masked carbenes in coupling reactions with sulfonic acids under photochemical conditions. Varieties of coupling partners including aminophosphonates and sulfonic acids are well tolerated in this reaction affording unknown sulfonate esters in good yields. The strategy of this new diazo compound provides a valuable vista for the construction of functionalized difluoromethylene phosphonates and transformations of fluoro diazo compounds.


 Please wait while we load your content...
Please wait while we load your content...