A hydrogen evolution catalyst lowering energy consumption in aluminum anodization†
Abstract
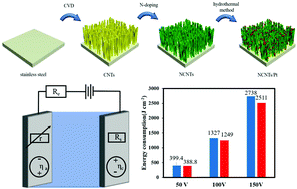
Aluminum anodization process requires a lot of electrical energy for the migration of ions in a barrier-type oxide film, in which electrode polarization leads to massive energy consumption. The cathodes are considered to be modified with catalysts, reducing their hydrogen evolution reaction (HER) overpotential in order to improve its energy utilization efficiency. In this study, N-doped carbon nanotube (NCNT)-supported Pt nanoparticle (NCNTs/Pt) electrodes were prepared instead of the traditional stainless steel cathodes. Considering the neutral electrolyte used for the aluminum anodizing process, the NCNTs/Pt electrodes were tested in an ammonium adipate (pH = 6.8), exhibiting an overpotential of 125 mV at −10 mA cm−2 and a mass activity of 107.6 A gPt−1. The NCNTs/Pt electrodes were employed as cathodes to measure the aluminum anodization energy consumption, which enabled 9.04% reduction at a formation voltage of 150 V compared with stainless steel electrodes. This study provides a way for energy conservation in the aluminum anodization process, with the novel pathway altering the cathodic electrode using carbon-based noble metals as efficient catalysts.
- This article is part of the themed collection: 2021 Inorganic Chemistry Frontiers HOT articles


 Please wait while we load your content...
Please wait while we load your content...