Poly(para-phenylene) ionomer membranes: effect of methyl and trifluoromethyl substituents†
Abstract
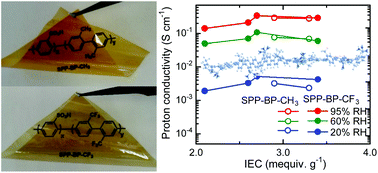
Sulfonated poly(para-phenylene)s with a high molecular weight and membrane forming capability were obtained by using methyl and trifluoromethyl substituents. The linearity of the polymer main chain decreased by introducing these substituents; the persistence length (lp, index of linearity, distance required for a polymer chain to bend by 90° on average) of homopolymers for 2,2′-dimethyl-1,1′-biphenyl (BP-CH3) and 2,2′-bis(trifluoromethyl)-1,1′-biphenyl (BP-CF3) was ca.350.6 nm and 87.7 nm, respectively, estimated by numerically averaging backbone conformations. Copolymers with sulfo-para-phenylene, SPP-BP-CH3 and SPP-BP-CF3, were obtained with a high molecular weight (Mn = 28–30 kDa and Mw = 88–100 kDa for SPP-BP-CH3 and Mn = 49–149 kDa and Mw = 161–316 kDa for SPP-BP-CF3, respectively) to provide flexible membranes by casting from the solution. Despite the more hydrophobic nature of the substituents, SPP-BP-CF3 membranes showed higher water uptake and proton conductivity than SPP-BP-CH3 membranes with comparable ion exchange capacity (IEC). SPP-BP-CF3 membranes showed slightly higher maximum strain (2.9–5.2%) than SPP-BP-CH3 membranes (1.1–2.1%), leading to a higher rupture energy as expected from the smaller persistence length of BP-CF3 homopolymers. While SPP-BP-CH3 decomposed under harsh oxidative conditions, SPP-BP-CF3 was more oxidatively stable and exhibited negligible changes in the weight, molecular weight, molecular structure and membrane properties (proton conductivity, mechanical properties, etc.).


 Please wait while we load your content...
Please wait while we load your content...