Does the oxa-Michael reaction of 2-trifluoromethacrylic acid lead to fluorinated polyesters?†
Abstract
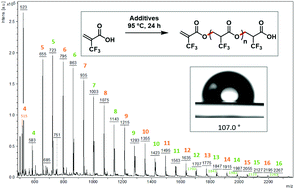
2-Trifluoromethacrylic acid (MAF) is a peculiar fluorinated functional monomer. Though it is known to polymerise via C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bond cleavage under anionic initiation, its oxa-Michael addition polymerisation afforded polyesters bearing trifluoromethyl side groups. This oxa-Michael addition was attempted in the presence of radical (peroxides), basic (amines and phosphines) and acidic (AlCl3, triflic acid, and p-toluenesulfonic acid) reactants and catalysts which led to the formation of oligomers with various yields (up to 57%) and molar masses (up to 2200 g mol−1). The obtained polyesters were characterised by GC/MS, IR, 19F NMR, 1H NMR and 13C NMR spectroscopy techniques and MALDI-TOF, and their thermal properties were determined by thermogravimetric analysis and differential scanning calorimetry. Triphenylphosphine and piperidine led to the highest degree of polymerization DPn (up to the 16th adduct) with a degradation temperature of about 200 °C. The glass transition temperatures (Tg) were −59 and −46 °C for the MAF bisadduct (diMAF) and oligoMAF, respectively. Such oligomers were hydrophobic, evidenced by a water contact angle measurement of 107°, showing that the contribution of the trifluoromethyl group was higher than that of the ester function.
C bond cleavage under anionic initiation, its oxa-Michael addition polymerisation afforded polyesters bearing trifluoromethyl side groups. This oxa-Michael addition was attempted in the presence of radical (peroxides), basic (amines and phosphines) and acidic (AlCl3, triflic acid, and p-toluenesulfonic acid) reactants and catalysts which led to the formation of oligomers with various yields (up to 57%) and molar masses (up to 2200 g mol−1). The obtained polyesters were characterised by GC/MS, IR, 19F NMR, 1H NMR and 13C NMR spectroscopy techniques and MALDI-TOF, and their thermal properties were determined by thermogravimetric analysis and differential scanning calorimetry. Triphenylphosphine and piperidine led to the highest degree of polymerization DPn (up to the 16th adduct) with a degradation temperature of about 200 °C. The glass transition temperatures (Tg) were −59 and −46 °C for the MAF bisadduct (diMAF) and oligoMAF, respectively. Such oligomers were hydrophobic, evidenced by a water contact angle measurement of 107°, showing that the contribution of the trifluoromethyl group was higher than that of the ester function.


 Please wait while we load your content...
Please wait while we load your content...