Porous silsesquioxane cage and porphyrin nanocomposites: sensing and adsorption for heavy metals and anions†
Abstract
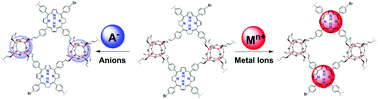
A porous silsesquioxane cage/porphyrin nanocomposite has been designed as a dual fluorescent probe for the sensing and adsorption of heavy metal ions and anions. The hybrid fluorescent probe (TPPSQ) has been synthesized via cross-linked polymerization between octavinylsilsesquioxane (SQ) and 5,10,15,20-tetrakis-(4-bromophenyl)porphyrin (TPP). Solid-state 29Si and 13C MAS-NMR, FTIR, and energy-dispersive X-ray (EDX) analysis confirmed the successful incorporation of TPP units within the SQ-based network. BET analysis and TGA analysis of the fluorescent nanocomposite revealed that the material possesses excellent thermal stability and a high surface area in the range of 433 m2 g−1, along with bimodal micropores, with a size distribution centered at 0.64 nm and 1.26 nm, and mesopores, with a size distribution centered at 2–3 nm. The incorporation of TPP units into the polymeric network makes the TPPSQ polymer fluorescent (λex = 420 nm, λem = 650 nm) in different media, which is crucial for chemosensing applications. The formation of excimers of TPP units within the polymeric network of TPPSQ plays a pivotal role in selective anion sensing, whereas the interaction capabilities of freebase TPP units with metal ions play a crucial role in cation sensing. Upon the addition of F− ions, TPPSQ showed F−-induced ‘turn-on’ detection due to interactions between the SQ cages and F− ions and the transformation of TPP excimers to monomers. On the other hand, after the addition of Hg2+ ions, TPPSQ showed instant ‘turn-off’ detection because, after the binding of TPP units with Hg2+ ions within the polymeric network, the material undertook a non-radiative relaxation pathway from the excited to the ground state. It is noteworthy that the material is capable of instantly detecting Hg2+ ions in an aqueous medium (DMSO/water ratio of 1 : 7), making it a potential candidate to act as a Hg2+ ion sensor in the real world. This porous nanocomposite (TPPSQ) is capable of adsorbing more than 90% of fluoride ions from solution within 60 min, whereas it can adsorb 76% of CN− ions from solution within 60 min. The nanoporous polymer is also capable of adsorbing 90% of Cu2+ and 88% of Hg2+ from solution within 60 min. The synthesized polymer TPPSQ has shown its excellent efficacy as a reusable fluoride- and Hg2+-ion sensor and as an adsorbent.


 Please wait while we load your content...
Please wait while we load your content...