Controlled ring-opening polymerization of N-(3-tert-butoxy-3-oxopropyl) glycine derived N-carboxyanhydrides towards well-defined peptoid-based polyacids†
Abstract
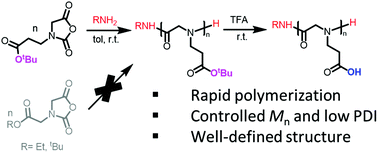
Polypeptoids bearing carboxylic acid groups on the N-substituent are useful building blocks for the construction of peptidomimetic supramolecular assemblies with stimuli-responsive properties. Towards this end, N-(3-tert-butoxy-3-oxopropyl) glycine derived N-carboxyanhydride (tBuO2Pr-NCA) has been successfully synthesized and polymerized using primary amine initiators to produce the corresponding poly(N-(3-tert-butoxy-3-oxopropyl) glycine) with molecular weights (Mn) of 5.6–59 kg mol−1 and a narrow molecular weight distribution (PDI = 1.003–1.026). The polymerization was shown to proceed in a controlled manner, evidenced by the good agreement of the experimental molecular weight (Mn) with theoretical values and narrow molecular weight distribution in a wide range of monomer-to-initiator ratios ([M]0 : [I]0 = 25 : 1–200 : 1), the linear increase of Mn with conversion and the second-order polymerization kinetics. The cloaked carboxyl groups on the poly(N-(3-tert-butoxy-3-oxopropyl) glycine) can be readily unveiled in mild acidic conditions to yield the poly(N-(2-carboxyethyl) glycine), a structural mimic of poly(glutamic acid). The poly(N-(2-carboxyethyl) glycine) polymer is a weak polyelectrolyte whose hydrodynamic size in water can be controlled by the solution pH.
- This article is part of the themed collection: Polymer Chemistry Pioneering Investigators 2021


 Please wait while we load your content...
Please wait while we load your content...