Fe/S-Catalyzed synthesis of 2-benzoylbenzoxazoles and 2-quinolylbenzoxazoles via redox condensation of o-nitrophenols with acetophenones and methylquinolines†
Abstract
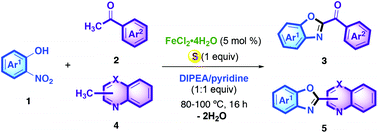
An Fe/S catalyst generated in situ from FeCl2·4H2O and elemental sulfur S8 in the presence of a tertiary amine as a base was found to catalyze efficiently a 6e− redox condensation of o-nitrophenols with acetophenones and methylquinolines. The condensed products 2-benzoylbenzoxazoles and 2-quinolylbenzoxazoles were obtained in reasonable yields with water as the only byproduct at a temperature as low as 80 °C.
- This article is part of the themed collection: Catalysis & biocatalysis in OBC


 Please wait while we load your content...
Please wait while we load your content...