Wavelength dependent photochemistry of BODIPY–phenols and their applications in the fluorescent labeling of proteins†
Abstract
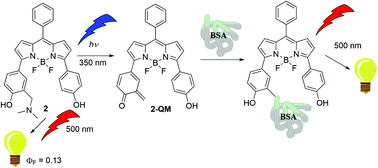
A series of BODIPY dyes were synthesized, that were at the 3, or 3 and 5 positions, substituted by photochemically reactive quinone methide (QM) precursor moieties. Fluorescence properties of the molecules were investigated and we demonstrated that the molecules undergo wavelength dependent photochemistry. Photodeamination to deliver QMs takes place only upon excitation to higher excited singlet states, showing unusual anti-Kasha photochemical reactivity. The findings were corroborated by TD-DFT computations. Laser flash photolysis experiments could not reveal QMs due to the low efficiency of their formation, but enabled the detection of phenoxyl radicals. The applicability of the molecules for the fluorescent labeling of bovine serum albumin as a model protein upon photoexcitation at 350 nm was demonstrated.
- This article is part of the themed collection: Mechanistic, computational & physical organic chemistry in OBC


 Please wait while we load your content...
Please wait while we load your content...