Design, synthesis, and evaluation of positron emission tomography/fluorescence dual imaging probes for targeting facilitated glucose transporter 1 (GLUT1)†
Abstract
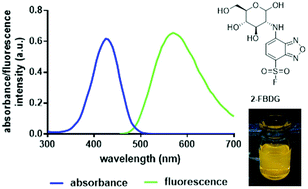
Increased energy metabolism followed by enhanced glucose consumption is a hallmark of cancer. Most cancer cells show overexpression of facilitated hexose transporter GLUT1, including breast cancer. GLUT1 is the main transporter for 2-deoxy-2-[18F]fluoro-D-glucose (2-[18F]FDG), the gold standard of positron emission tomography (PET) imaging in oncology. The present study's goal was to develop novel glucose-based dual imaging probes for their use in tandem PET and fluorescence (Fl) imaging. A glucosamine scaffold tagged with a fluorophore and an 18F-label should confer selectivity to GLUT1. Out of five different compounds, 2-deoxy-2-((7-sulfonylfluoro-2,1,3-benzoxadiazol-4-yl)amino)-D-glucose (2-FBDG) possessed favorable fluorescent properties and a similar potency as 2-deoxy-2-((7-nitro-2,1,3-benzoxadiazol-4-yl)amino)-D-glucose (2-NBDG) in competing for GLUT1 transport against 2-[18F]FDG in breast cancer cells. Radiolabeling with 18F was achieved through the synthesis of prosthetic group 7-fluoro-2,1,3-benzoxadiazole-4-sulfonyl [18F]fluoride ([18F]FBDF) followed by the reaction with glucosamine. The radiotracer was finally analyzed in vivo in a breast cancer xenograft model and compared to 2-[18F]FDG. Despite favourable in vitro fluorescence imaging properties, 2-[18F]FBDG was found to lack metabolic stability in vivo, resulting in radiodefluorination. Glucose-based 2-[18F]FBDG represents a novel dual-probe for GLUT1 imaging using FI and PET with the potential for further structural optimization for improved metabolic stability in vivo.
- This article is part of the themed collection: Chemical Biology in OBC


 Please wait while we load your content...
Please wait while we load your content...