The implication of adsorption preferences of ions and surfactants on the shape control of gold nanoparticles: a microscopic, atomistic perspective†
Abstract
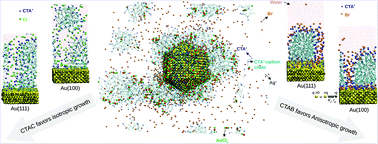
Shape modulation of nanoparticles is crucial for their tailored applications; however, it depends on surfactants, ions, reactants, and other additives present in the growth solution. Here we dissect the role of surfactants, their counterions (halide ions), silver ions, and gold reactant in gold nanoparticle anisotropic growth using polarizable surfaces and nanoseed molecular dynamics simulation models. Our planar surface models predict a 14%–16% increment in cetyltrimethylammonium bromide (CTAB) coverage on Au(111) and Au(100) due to the surface polarization effect. The CTAB micelle adsorbs compactly similar to that observed on non-polarizable surfaces. The cetyltrimethylammonium chloride (CTAC) micelle remains in solution leaving the polarizable gold surfaces unprotected, similar to that observed with the non-polarizable surfaces, which favors isotropic growth. The cetyltrimethylammonium iodide (CTAI) micelle adsorbs with higher surface densities than CTAB on all the surfaces. The surface polarizable penta-twinned nanoseed model predicts the total surface coverage of the cetyltrimethylammonium cation (CTA+), Br− and Ag+ to be around two times higher on the side as compared to the tip of the nanoseed, leading to a 2.6 times higher initial rate of adsorption of AuCl2− on the tip than on the side. Predicted CTA+ surface densities on the tip and the side of the nanoseed are consistent with experimental results. Our simulations explain the growth mechanism of anisotropic nanoparticles and the microscopic origin of their controlled shapes.


 Please wait while we load your content...
Please wait while we load your content...