Quantifying the force in flow-cell based single-molecule stretching experiments†
Abstract
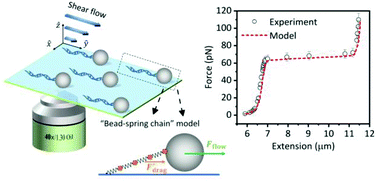
The flow-cell based single-molecule manipulation technique has found many applications in the study of DNA mechanics and protein–DNA interactions. However, the force in these experiments has not been fully characterized and is usually limited to a moderate force regime (<25 pN). In this work, using the “tethered-bead” assay, the hydrodynamic drag of DNA has been quantitatively evaluated based on a “bead-spring chain” model. The force derived from the Brownian motion of the bead thus contains both contributions from this equivalent hydrodynamic drag of DNA and the pulling force from the tethered bead. Next, using flow-cell based DNA pulling experiments, the linear relationship between the flow rate and total hydrodynamic force on the bead-DNA system has been demonstrated to be valid over a wide force range (0–110 pN). Consequently, the force can be directly converted from the flow rate by a linear factor that can be calibrated either by the bead's Brownian motion at low flow rates or using DNA overstretching transition. Furthermore, the hydrodynamic force and torque due to the shear flow on the bead as well as the equivalent stretching force on DNA are calculated based on theoretical models with the hydrodynamic drag on DNA also considered. The calculated force–extension curves show a good agreement with the measured ones. These results offer important insights into the force in flow-cell based single-molecule stretching experiments and provide a foundation for establishing flow-cells as a simple, low-cost, yet flexible and precise tool for single-molecule force measurements over a wide force range.


 Please wait while we load your content...
Please wait while we load your content...