Surface chemistry in calcium capped carbon quantum dots†
Abstract
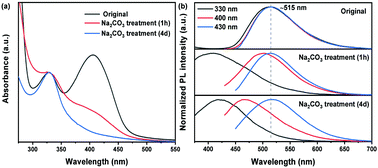
Colloidal carbon quantum dots (C-dots) have attracted a lot of attention because of their excellent optical properties for various types of applications. Due to the complicated structure of C-dots, the photoluminescence (PL) mechanism of C-dots is still unclear. In particular, it is still a big challenge to understand well the surface chemistry of C-dots. In this work, we used a vacuum-heating approach to produce high-quality C-dots. With different purification procedures, the surface chemistry of C-dots can be well-controlled. Removal of Ca2+ by Na2CO3 led to the disappearance of the absorption at 405 nm and a decrease of the quantum yield. In addition, the Na2CO3 treated C-dots exhibited an excitation-dependent PL behavior. These results confirmed that Ca2+ can interact with the surface functional group of C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O of the C-dots, forming a stable structure surrounding the C-dot core, which contributed to a high quantum yield (QY) of 65%, excitation-independent PL behavior and absorption at 405 nm. Furthermore, the PL of the C-dots is strongly dependent on the pH, indicating that the Ca2+ capped C-dots could be used as pH indicators. Our finding provides clear evidence for the surface-chemistry dependent PL behavior of C-dots.
O of the C-dots, forming a stable structure surrounding the C-dot core, which contributed to a high quantum yield (QY) of 65%, excitation-independent PL behavior and absorption at 405 nm. Furthermore, the PL of the C-dots is strongly dependent on the pH, indicating that the Ca2+ capped C-dots could be used as pH indicators. Our finding provides clear evidence for the surface-chemistry dependent PL behavior of C-dots.


 Please wait while we load your content...
Please wait while we load your content...