Improvement of alkali metal ion batteries via interlayer engineering of anodes: from graphite to graphene†
Abstract
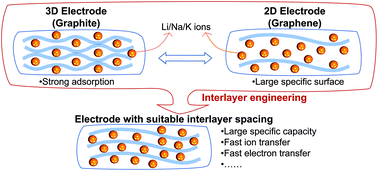
Interlayer engineering of graphite anodes in alkali metal ion (M = Li, Na, and K) batteries is carried out based on the first-principles calculations. By increasing the interlayer spacing of graphite, the specific capacity of Li or Na does not increase while that of K increases continuously (from 279 mA h g−1 at the equilibrium interlayer spacing to 1396 mA h g−1 at the interlayer spacing of 20.0 Å). As the interlayer spacing increases, the electrostatic potential of graphite becomes smoother, and the ability to buffer the electrostatic potential fluctuation becomes poorer in M ions. These two effects jointly lead to minima of the diffusion barrier of M ions on graphite (0.01–0.05 eV), instead of strictly monotonous declines with the increasing interlayer spacing. To perform the interlayer engineering of anode candidates more efficiently, a set of high-throughput programs has been developed and can be easily applied to other systems. Our research has guiding significance for achieving the optimal effect in interlayer engineering experimentally.
- This article is part of the themed collection: 2021 Nanoscale HOT Article Collection


 Please wait while we load your content...
Please wait while we load your content...