Synthetic strategies for the ibophyllidine alkaloids
Abstract
Covering: 1975–2020
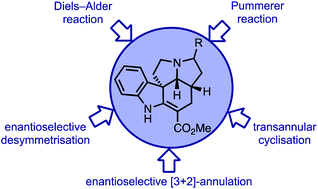
The ibophyllidine alkaloids are unique pyrroloindole alkaloids exhibiting a five-membered D-ring in contrast to the six-membered D-ring of the more common Aspidosperma and Strychnos alkaloids. This structural feature has made them sought-after targets for organic chemists as well as for the elucidation of their biosynthesis. Beginning with the first and eponymous member ibophyllidine, isolation and structure determination is discussed. The main focus of this review are the diverse chemical approaches towards the ibophyllidines in context with their respective biosynthesis. The often employed Diels–Alder reaction strategy, two other named reaction-based strategies and the most recent enantioselective strategies are presented and compared.


 Please wait while we load your content...
Please wait while we load your content...