Discovery of new indole-based 1,2,4-triazole derivatives as potent tubulin polymerization inhibitors with anticancer activity†
Abstract
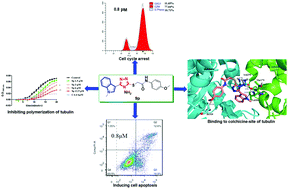
Thirty-six novel indole-based 1,2,4-triazole derivatives were designed and synthesized through the molecular hybrid strategy. The bioassay results revealed that 9p displayed excellent antiproliferative efficacies in the nanomolar range against HeLa cells. Importantly, the compound exhibited no obvious cytotoxic activity (IC50 > 100 μM) toward HEK-293, a normal human embryonic kidney cell line. Mechanism analysis indicated that 9p significantly arrested the cell cycle at the G2/M phase and induced apoptosis in HeLa cells in a dose-dependent manner. Further evidence demonstrated that the promising compound effectively inhibited tubulin polymerization with an IC50 value of 8.3 μM, and molecular docking studies revealed that 9p well occupied the colchicine-site in tubulin. The present study highlights that indole–triazole hybrids might be used as a promising scaffold to develop novel tubulin polymerization inhibitors for cancer treatment.


 Please wait while we load your content...
Please wait while we load your content...