Radical sensitivity and selectivity in the electrochemical sensing of cadmium ions in water by polyaniline-derived nitrogen-doped graphene quantum dots†
Abstract
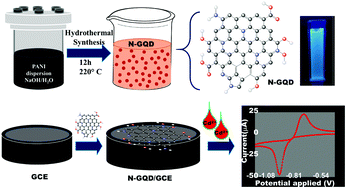
In the present work, the sensing capability of nitrogen-doped graphene quantum dots (N-GQDs) was explored for the first time toward hazardous heavy metal ions and they were found to be able to selectively detect cadmium ions (Cd(II)). Cd(II) is one of the hazardous heavy metal ions on the World Health Organization's list of 10 chemicals of major public health concern. The wide use of Cd(II) in industrial applications and its acute and chronic effects on living beings make the sensing of Cd(II) critical. The aromatic N-GQDs, synthesized here through a hydrothermal route using the single precursor polyaniline exhibited a 25- and 6-fold increase in the current response compared to that of a glassy carbon electrode (GCE) and GQD/GCE, respectively. It showed an excellent and exemplary selectivity toward Cd(II), with almost a nil current response for other interfering metal ions. The LOD value of 1.0 × 10−5 ppb, i.e., in parts per trillion, by N-GQDs is the lowest value ever reported, which was 3–4 orders of magnitude lower than the previously reported values. The LOD was extended to the ultra-high sensitivity level of 1 × 10−7 ppb, i.e., in parts per quadrillion, using a pre-reduction step. The LDR (0.001–112 000 ppb) and LOD values satisfactorily met the sensitivity requirement for detecting Cd(II) within the safe limit specified by the U.S. Environmental Protection Agency (EPA) of ≤5 ppb. The sensing performance of N-GQD/GCE was tested against real environmental samples spiked with Cd(II), such as groundwater, seawater, and wastewater, and was found to perform efficiently.


 Please wait while we load your content...
Please wait while we load your content...