Nano-manganese oxide and reduced graphene oxide-incorporated polyacrylonitrile fiber mats as an electrode material for capacitive deionization (CDI) technology†
Abstract
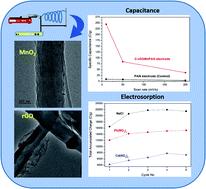
Capacitive deionization (CDI) is a trending water desalination method during which the impurity ions in water can be removed by electrosorption. In this study, nano-manganese dioxide (MnO2) and reduced graphene oxide (rGO)-doped polyacrylonitrile (PAN) composite fibers are fabricated using an electrospinning technique. The incorporation of both MnO2 and rGO nanomaterials in the synthesized fibers was confirmed by transmission electron microscopy (TEM), scanning electron microscopy (SEM), and energy dispersive X-ray spectroscopy (EDX). The electrochemical characteristics of electrode materials were examined using cyclic voltammetry (CV), electrochemical impedance spectroscopy (EIS) and constant current charge–discharge cycles (CCCDs). The specific capacitance of the PAN electrode increased with increasing MnO2 and rGO contents as well as when thermally treated at 280 °C. Thermally treated composite fibers with 17% (w/w) MnO2 and 1% (w/w) rGO (C-rGOMnPAN) were observed to have the best electrochemical performance, with a specific capacitance of 244 F g−1 at a 10 mV s−1 scan rate. The electrode system was used to study the removal of sodium chloride (NaCl), cadmium (Cd2+) and lead (Pb2+) ions. Results indicated that NaCl showed the highest electrosorption (20 472 C g−1) compared to two heavy metal salts (14 260 C g−1 for Pb2+ and 6265 C g−1 for Cd2+), which is most likely to be due to the ease of mass transfer of lighter Na+ and Cl− ions; When compared, Pb2+ ions tend to show more electrosorption on these fibers than Cd2+ ions. Also, the C-rGOMnPAN electrode system is shown to work with 95% regeneration efficiency when 100 ppm NaCl is used as the electrolyte. Hence, it is clear that the novel binder-free, electrospun C-rGOMnPAN electrodes have the potential to be used in salt removal and also for the heavy metal removal applications of water purification.
- This article is part of the themed collection: Popular Advances


 Please wait while we load your content...
Please wait while we load your content...