Development, structure and mechanics of a synthetic E. coli outer membrane model†
Abstract
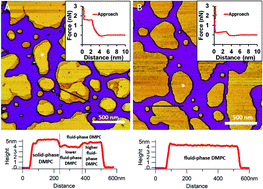
The outer membrane (OM) of Gram-negative bacteria is a complex asymmetric bilayer containing lipids, lipopolysaccharides (LPS) and proteins. While it is a mechanical and chemical barrier, it is also the primary surface of bacterial recognition processes that involve infection by and of the bacterium. Uncovering the mechanisms of these biological functions has been hampered by the lack of suitable model systems. Here we report the step-by-step assembly of a synthetic OM model from its fundamental components. To enable the efficient formation of a supported lipid bilayer at room temperature, dimyristoyl-phosphocholine (DMPC) was used as the lipid component to which we progressively added LPS and OM proteins. The assembled system enabled us to explore the contribution of the molecular components to the topographical structure and stability of the OM. We found that LPS prefers solid-state membrane regions and forms stable vesicles in the presence of divalent cations. LPS can gradually separate from DMPC membranes to form independent vesicles, pointing at the dynamic nature of the lipid-LPS system. The addition of OM proteins from E. coli and saturating levels of LPS to DMPC liposomes resulted in a thicker and more stable bilayer the surface of which displayed a nanoscale texture formed of parallel, curved, long (>500 nm) stripes spaced apart with a 15 nm periodicity. The synthetic membrane may facilitate the investigation of binding and recognition processes on the surface of Gram-negative bacteria.
- This article is part of the themed collection: Editor’s Choice: Recent breakthroughs in nanobiotechnology


 Please wait while we load your content...
Please wait while we load your content...