Replacement of oxygen with sulfur on the furanose ring of cyclic dinucleotides enhances the immunostimulatory effect via STING activation†
Abstract
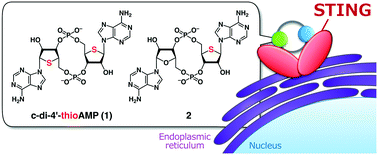
Cyclic dinucleotides (CDNs) are secondary messengers composed of two purine nucleotides linked via two phosphodiester linkages: c-di-GMP, c-di-AMP, 3′,3′-cGAMP, and 2′,3′-cGAMP. CDNs activate the stimulator of interferon genes (STING) and trigger immune responses in mammalian species. CDNs are thus fascinating molecules as drug candidates, and chemically stable CDN analogues that act as STING agonists are highly desired at present. We herein report the practical synthesis of 4′-thiomodified c-di-AMP analogues, which have sulfur atoms at the 4′-position on the furanose ring instead of oxygen atoms, using simple phosphoramidite chemistry. The resulting 4′-thiomodified c-di-AMP analogues acted as potent STING agonists with long-term activity. Our results show that replacing O4′ on CDNs with sulfur can lead to enhanced immunostimulatory effects via STING activation.
- This article is part of the themed collection: Emerging Investigators


 Please wait while we load your content...
Please wait while we load your content...