A selective detection of nanomolar-range noxious anions in water by a luminescent metal–organic framework†
Abstract
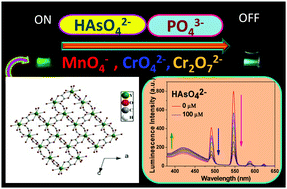
A new metal–organic framework (MOF) showed excellent recognition ability toward five toxic oxo-anions, namely arsenate (HAsO42−), phosphate (PO43−), permanganate (MnO4−), chromate (CrO42−), and dichromate (Cr2O72−) in aqueous medium upon irradiation with UV light. The MOF [Y(tp)(ox)0.5(H2O)2]·H2O, 1 and a 20% Tb-doped MOF [Y0.8Tb0.2(tp)(ox)0.5(H2O)2]·H2O, 1a were synthesized by the self-assembly of Y(III)/Tb(III), terephthalic acid (H2tp), and oxalic acid (H2ox) under hydrothermal conditions. The crystal structure of 1 demonstrated three-dimensional structures with water occupying a one-dimensional channel. Upon UV light exposure, an aqueous suspension of 1a exhibited strong green luminescence. This luminescence showed a colour change when water was contaminated by the said anions. Several other interfering anions were also tested to establish its selectivity as a chemosensor toward the aforementioned anions. The estimated limit of detection (LOD) of 1a for these anions was 41.7, 33.5, 19.6, 40.8, and 43.9 nM for HAsO42−, PO43−, MnO4−, CrO42−, and Cr2O72−, respectively, which were significantly lower than the levels recommended for these anions in the aqueous environment. The root of the luminescence quenching of 1a in the presence of such a low concentration of aforementioned anions was due to the interruption of the sensitization process of the Tb centre. Anions HAsO42−, PO43−, and MnO4− entered the channel of MOF and interacted with Y3+/Tb3+ centres at the molecular-level, which hampered the energy flow from the ligand to the metal centre through a weakening of Tb–O bonds of carboxylates, resulting in a quenching of the Tb centre emission with a concomitant enhancement of the ligand centre emission. Additionally, in the case of the MnO4− ion, an inner filter effect also contributed to the quenching of the Tb centre emission, but to a smaller extent. For CrO42− and Cr2O72− anions, the inner filter effect is the key mechanism for the luminescence intensity reduction. Besides, these analytes could also reduce the energy flow from the ligand to the lanthanide ion, which led to the luminescence quenching.
- This article is part of the themed collection: Fluorescent and Luminescent Materials


 Please wait while we load your content...
Please wait while we load your content...