Engineering of a functional pancreatic acinus with reprogrammed cancer cells by induced PTF1a expression†
Abstract
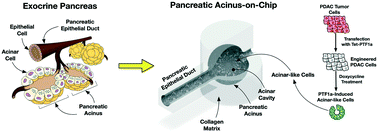
A pancreatic acinus is a functional unit of the exocrine pancreas producing digest enzymes. Its pathobiology is crucial to pancreatic diseases including pancreatitis and pancreatic cancer, which can initiate from pancreatic acini. However, research on pancreatic acini has been significantly hampered due to the difficulty of culturing normal acinar cells in vitro. In this study, an in vitro model of the normal acinus, named pancreatic acinus-on-chip (PAC), is developed using reprogrammed pancreatic cancer cells. The developed model is a microfluidic platform with an epithelial duct and acinar sac geometry microfabricated by a newly developed two-step controlled “viscous-fingering” technique. In this model, human pancreatic cancer cells, Panc-1, reprogrammed to revert to the normal state upon induction of PTF1a gene expression, are cultured. Bioinformatic analyses suggest that, upon induced PTF1a expression, Panc-1 cells transition into a more normal and differentiated acinar phenotype. The microanatomy and exocrine functions of the model are characterized to confirm the normal acinus phenotypes. The developed model provides a new and reliable testbed to study the initiation and progression of pancreatic cancers.


 Please wait while we load your content...
Please wait while we load your content...