Direct isolation of circulating extracellular vesicles from blood for vascular risk profiling in type 2 diabetes mellitus†
Abstract
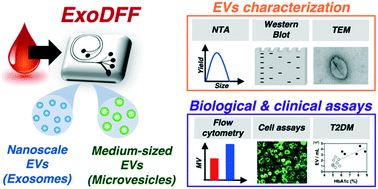
Extracellular vesicles (EVs) are key mediators of communication among cells, and clinical utilities of EVs-based biomarkers remain limited due to difficulties in isolating EVs from whole blood reliably. We report a novel inertial-based microfluidic platform for direct isolation of nanoscale EVs (exosomes, 50 to 200 nm) and medium-sized EVs (microvesicles, 200 nm to 1 μm) from blood with high efficiency (three-fold increase in EV yield compared to ultracentrifugation). In a pilot clinical study of healthy (n = 5) and type 2 diabetes mellitus (T2DM, n = 9) subjects, we detected higher EV levels in T2DM patients (P < 0.05), and identified a subset of “high-risk” T2DM subjects with abnormally high (∼10-fold to 50-fold) amounts of platelet (CD41a+) or leukocyte-derived (CD45+) EVs. Our in vitro endothelial cell assay further revealed that EVs from “high-risk” T2DM subjects induced significantly higher vascular inflammation (ICAM-1 expression) (P < 0.05) as compared to healthy and non-“high-risk” T2DM subjects, reflecting a pro-inflammatory phenotype. Overall, the EV isolation tool is scalable, and requires less manual labour, cost and processing time. This enables further development of EV-based diagnostics, whereby a combined immunological and functional phenotyping strategy can potentially be used for rapid vascular risk stratification in T2DM.
- This article is part of the themed collections: Microfluidics for hematology and Lab on a Chip HOT Articles 2021


 Please wait while we load your content...
Please wait while we load your content...