CO2-tuned highly selective reduction of formamides to the corresponding methylamines†
Abstract
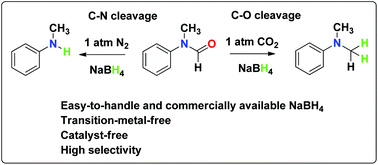
We herein describe an efficient, CO2-tuned and highly selective C–O bond cleavage of N-methylated formanilides. With easy-to-handle and commercially available NaBH4 as the reductant, a variety of formanilides could be turned into the desired tertiary amines in moderate to excellent yields. The role of CO2 has been investigated in detail, and the mechanism is proposed on the basis of experiments.


 Please wait while we load your content...
Please wait while we load your content...