Ultrasound-assisted extraction of metals from Lithium-ion batteries using natural organic acids†
Abstract
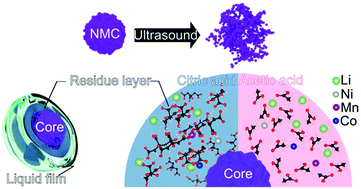
An ultrasound-assisted extraction (leaching) method of valuable metals from discarded lithium-ion batteries (LiBs) is reported. Mild organic citric or acetic acids were used as leaching agents for a more environmentally-friendly recovery of the lithium, nickel, cobalt, and manganese from the discharged and crushed lithium nickel-manganese-cobalt oxide (NMC) LiBs. The extraction was performed with the presence/absence of continuous ultrasound (US) energy supplied by a 110 W ultrasonic bath. The effect of temperature (30–70 °C), reducing agent concentration (H2O2: 0–2.0 vol%), as well as choice of specific acid on the metal dissolution were investigated. The US leaching decreased the leaching time by more than 50% and improved the leached percentage of Li, Mn, Co, and Ni due to the local heat and improved mass transfer between solid and liquid interfaces in the process. The X-ray diffraction results of residues from the US leaching further confirmed an improved dissolution of the crushed layered NMC structure, resulting in the significant improvement of the leached amounts of the valuable metals. Furthermore, it is demonstrated that using citric acid eliminated the need of additional reducing agents and suppressed the dissolution of copper (Cu) due to its inhibitor effect on the Cu surface, i.e. compared with using acetic acid as leaching reagent. Overall, it is shown that recovery of the battery metals can be facilitated and carried out in a more energy-efficient manner at low temperatures (50 °C) using ultrasound to improve metal ions mass transportation in the residue layers of the NMC during the organic acid leaching.


 Please wait while we load your content...
Please wait while we load your content...