Visible light initiated oxidative coupling of alcohols and o-phenylenediamines to synthesize benzimidazoles over MIL-101(Fe) promoted by plasmonic Au†
Abstract
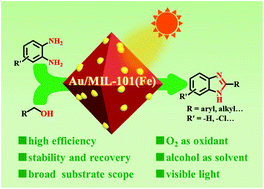
The use of visible light to initiate one-pot synergistic/cascade reactions is a green and energy saving strategy. In this manuscript, we report that MIL-101(Fe) can act as a multifunctional catalyst to realize the oxidative condensation between o-phenylenediamines and alcohols to synthesize benzimidazoles under visible light. The deposition of plasmonic Au nanoparticles (Au NPs) on MIL-101(Fe) led to significantly improved activity. Both controlled experiments and electron spin resonance (ESR) results revealed that the production of benzimidazoles from o-phenylenediamines and alcohols involves three sequential steps, i.e., the oxidative dehydrogenation of alcohols to produce aldehydes, the condensation between o-phenylenediamines and the aldehydes to produce Schiff bases and their oxidation to form benzimidazoles, via a superoxide radical (O2˙−)-mediated pathway. The promoting effect of plasmonic Au NPs in this reaction can be ascribed to the effective transfer of the surface plasmon resonance (SPR)-excited hot electrons to the lowest unoccupied molecular orbital (LUMO) of MIL-101(Fe), which led to the generation of more active O2˙− radicals. This study not only provides a green and sustainable way for the synthesis of benzimidazoles, but also highlights the great potential of using rationally designed plasmonic metal NP/MOF nanocomposites as multifunctional catalysts for light initiated one-pot tandem/cascade reactions.


 Please wait while we load your content...
Please wait while we load your content...