Bioactive peptide release and the absorption tracking of casein in the gastrointestinal digestion of rats†
Abstract
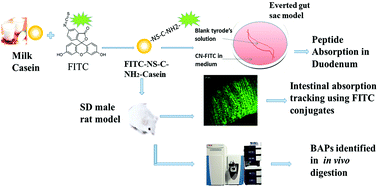
Bovine casein is considered as an important source of many bioactive peptides (BAPs), which can also be produced via in vitro simulated gastrointestinal hydrolysis. To perform their physiological functions, some active peptides need to pass through the intestinal epithelial barrier and keep their structural integrity after oral administration. Owing to the complexity of in vivo digestion and absorption, there have been few studies in this area. In this study, casein was labeled with FITC to trace its digestion and absorption in Sprague Dawley (SD) rats. Gastric juice, intestinal fluid, blood, and intestinal tissue samples were collected at different time-points for preservation and analysis after intragastric administration. The results showed that CN-FITC exhibited good labeling stability in the gastrointestinal digestive juice both in vivo and in vitro, suggesting its potential to be used for the detection and tracking of casein hydrolysate. After the intra-gastric administration of FITC, the diffusion rates of fluorescent substances in serum were much higher than in the CN-FITC group. The maximum peptide content in the CN-FITC group during intestinal digestion was achieved 2 h after administration, and electrophoretic analysis of the hydrolysate composition suggested that the molecular weights of the peptides were mainly concentrated in the range of 3.4–10 kDa. The hydrolyzed peptides from CN-FITC could be absorbed into the blood just 1 h after administration. Frozen sections of rat duodenal tissue were observed under a confocal laser scanning microscope, and they showed that the CN-FITC digested products were absorbed from villi to mucosa in the rat intestines, and the casein-hydrolyzed polypeptides were accumulated significantly in tissue samples 2 h after administration. The peptides were mainly absorbed in the duodenum on the basis of absorption experiments using an everted gut sac. After intestinal digestion for 2 h, peptides with weights less than 5 kDa were enriched and identified using LC-MS-MS, and they were found to be mainly derived from β-casein, containing potential angiotensin-I-converting enzyme inhibitory, antioxidant, dipeptidyl peptidase IV inhibitory, and morphine-like peptides. The peptides from casein hydrolysate were tracked entering the blood through the intestinal epithelial barrier in the form of complete fragments, and they might exert potential physiological activity in vivo.


 Please wait while we load your content...
Please wait while we load your content...