Isoleucine increases muscle mass through promoting myogenesis and intramyocellular fat deposition†
Abstract
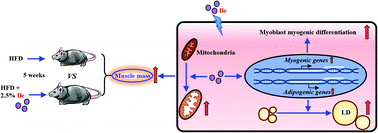
Isoleucine (Ile), as a branched-chain amino acid (BCAA), has a vital role in regulating body weight and muscle protein synthesis. However, the regulatory effect of Ile on muscle mass under high-fat diet (HFD) conditions and intramyocellular lipid deposition remains largely unclear. In this study, a feeding experiment with HFD with or without 25 g L−1 Ile was performed using 32 wild male C57BL/6J mice randomly divided into two groups. The results showed that Ile significantly increased both muscle and fat mass, as well as causing insulin resistance and meanwhile upregulating the levels of key adipogenic and myogenic proteins. More importantly, Ile damaged the mitochondrial function by vacuolation, swelling and cristae fracture in the gastrocnemius (GAS) and tibialis anterior (TA) with downregulation of mitochondrial function-related genes. Furthermore, Ile promoted myogenesis and more lipid droplet accumulation in myotubes. Compared with the control, the protein levels of myosin heavy chain (MyHC), myoblast determination protein 1 (MyoD), myogenin (MyoG), peroxisome proliferator-activated receptor gamma (PPARg) and fatty acid synthase (FAS) were upregulated in the Ile group, whereas the protein levels of adipose triglyceride lipase (ATGL) and lipoprotein lipase (LPL) were downregulated. Collectively, Ile increased muscle mass through myogenesis and intramyocellular lipid deposition. Our findings provide a new perspective for not only improving the lean juiciness of farm animals by increasing intramyocellular lipid accumulation, but also modulating myopathies under obesity.


 Please wait while we load your content...
Please wait while we load your content...