CO2 utilization in built environment via the PCO2 swing carbonation of alkaline solid wastes with different mineralogy
Abstract
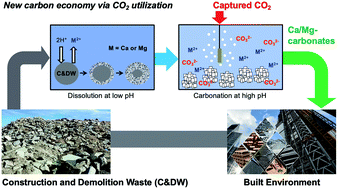
Carbon mineralization to solid carbonates is one of the reaction pathways that can not only utilize captured CO2 but also potentially store it in the long term. In this study, the dissolution and carbonation behaviors of alkaline solid wastes (i.e., waste concrete) was investigated. Concrete is one of the main contributors to a large carbon emission in the built environment. Thus, the upcycling of waste concrete via CO2 utilization has multifaceted environmental benefits including CO2 emission reduction, waste management and reduced mining. Unlike natural silicate minerals such as olivine and serpentine, alkaline solid wastes including waste concrete are highly reactive, and thus, their dissolution and carbonation behaviors vary significantly. Here, both conventional acid (e.g., hydrochloric acid) and less studied carbonic acid (i.e., CO2 saturated water) solvent systems were explored to extract Ca from concrete. Non-stoichiometric dissolution behaviors between Ca and Si were confirmed under far-from-equilibrium conditions (0.1 wt% slurry density), and the re-precipitation of the extracted Si was observed at near-equilibrium conditions (5 wt% slurry density), when the Ca extraction was performed at a controlled pH of 3. These experiments, with a wide range of slurry densities, provided valuable insight into Si re-precipitation phenomena and its effect on the mass transfer limitation during concrete dissolution. Next, the use of the partial pressure of CO2 for the pH swing carbon mineralization process was investigated for concrete, and the results were compared to those of Mg-bearing silicate minerals. In the PCO2 swing process, the extraction of Ca was significantly limited by the precipitation of the carbonate phase (i.e., calcite), since CO2 bubbling could not provide a low enough pH condition for concrete–water–CO2 systems. Thus, this study showed that the two-step carbon mineralization via PCO2 swing, that has been developed for Mg-bearing silicate minerals, may not be viable for highly reactive Ca-bearing silicate materials (e.g., concrete). The precipitated calcium carbonate (PCC) derived from waste concrete via a pH swing process showed very promising results with a high CO2 utilization potential as an upcycled construction material.
- This article is part of the themed collection: Carbon dioxide utilisation


 Please wait while we load your content...
Please wait while we load your content...