Improved photodegradation of anionic dyes using a complex graphitic carbon nitride and iron-based metal–organic framework material†
Abstract
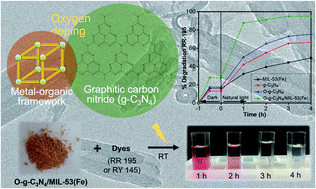
Introducing heterostructures to graphitic carbon nitrides (g-C3N4) can improve the activity of visible-light-driven catalysts for the efficient treatment of multiple toxic pollutants in water. Here, we report for the first time that a complex material can be constructed from oxygen-doped g-C3N4 and a MIL-53(Fe) metal–organic framework using facile hydrothermal synthesis and recycled polyethylene terephthalate from plastic waste. The novel multi-walled nanotube structure of the O-g-C3N4/MIL-53(Fe) composite, which enables the unique interfacial charge transfer at the heterojunction, showed an obvious enhancement in the separation efficiency of the photochemical electron–hole pairs. This resulted in a narrow bandgap energy (2.30 eV, compared to 2.55 eV in O-g-C3N4), high photocurrent intensity (0.17 mA cm−2, compared to 0.12 mA cm−2 and 0.09 mA cm−2 in MIL-53(Fe) and O-g-C3N4, respectively) and excellent catalytic performance in the photodegradation of anionic azo dyes (95% for RR 195 and 99% for RY 145 degraded after 4 h, and only a minor change in the efficiency observed after four consecutive tests). These results demonstrate the development of new catalysts made from waste feedstocks that show high stability, ease of fabrication and can operate in natural light for environmental remediation.
- This article is part of the themed collection: MOFs for energy and environmental applications


 Please wait while we load your content...
Please wait while we load your content...