Low-energy electron irradiation induced synthesis of molecular nanosheets: influence of the electron beam energy†
Abstract
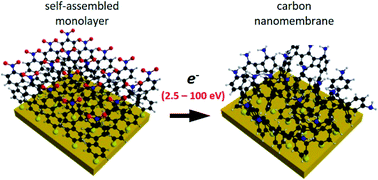
Aromatic self-assembled monolayers (SAMs) can be cross-linked into molecular nanosheets – carbon nanomembranes (CNMs) – via low-energy electron irradiation. Due to their favorable mechanical stability and tunable functional properties, they possess a high potential for various applications including nanosensors and separation membranes for osmosis or energy conversion devices. Despite this potential, the mechanistic details of the electron irradiation induced cross-linking process still need to be understood in more detail. Here, we studied the cross-linking of 4′-nitro-1,1′-biphenyl-4-thiol SAM on gold. The SAM samples were irradiated with different electron energies ranging from 2.5 to 100 eV in ultra-high vacuum and subsequently analysed by complementary techniques. We present results obtained via spectroscopy and microscopy characterization by high-resolution X-ray photoelectron spectroscopy (XPS), low-energy electron diffraction with micrometre sized electron beams (μLEED) and low-energy electron microscopy (LEEM). To demonstrate the formation of CNMs, the formed two-dimensional molecular materials were transferred onto grids and oxidized wafer and analyzed by optical, scanning electron microscopy (SEM) and atomic force microscopy (AFM). We found a strong energy dependence for the cross section for the cross-linking process, the rate of which decreases exponentially towards lower electron energies by about four orders of magnitude. We conduct a comparative analysis of the cross sections for the C–H bond scission via electron impact ionization and dissociative electron attachment and find that these different ionization mechanisms are responsible for the variation of the cross-linking cross section with electron energy.
- This article is part of the themed collection: Chemistry of 2-dimensional materials: beyond graphene


 Please wait while we load your content...
Please wait while we load your content...