Degradation of atrazine in the electrochemical LED-UV/Cl2 system: the role of ˙OH and Cl˙†
Abstract
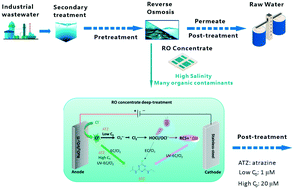
In this study, electrochemically generated free chlorine (EC/Cl2) was activated by UV275nm irradiation to degrade atrazine, a commonly used herbicide, investigating the degradation kinetics and mechanisms. The potential at a RuO2/IrO2–Ti anode was optimized as 1.5 V vs. Ag/AgCl through electrochemical measurement methods. The effects of atrazine initial concentration (C0) on the UV-EC/Cl2 and EC/Cl2 processes were evaluated. The atrazine degradation efficiency under low C0 conditions is surprisingly distinct from that under high C0 conditions. Under low C0 (1 μM) conditions, UV-EC/Cl2 degraded 60% atrazine at 60 min with ˙OH as the dominant species, while EC/Cl2 only removed 6% atrazine within 60 min. Under high C0 (20 μM) conditions, similar atrazine degradation efficiencies (about 90% within 180 min) were observed both in UV-EC/Cl2 and EC/Cl2. 50 μM nitrobenzene was used with 20 μM atrazine to conduct competition experiments, which confirmed the significant role of Cl˙ in atrazine degradation in the UV-EC/Cl2 and EC/Cl2 systems. During atrazine degradation by UV-EC/Cl2 and EC/Cl2, transformation products were compared according to high-resolution mass spectra, based on which possible degradation pathways were proposed. Synthetic reverse osmosis concentrate (ROC) was used to mimic the ROC from industrial wastewater and roughly evaluate the performances of UV-EC/Cl2 for the degradation of emerging contaminants, including atrazine, ibuprofen, carbamazepine, and diclofenac. Overall, EC/Cl2 can be effective at optimized anodic potential for the degradation of contaminants that are at high C0 and quickly react with Cl˙, while UV-EC/Cl2 is able to generate ˙OH and reactive chlorine species to efficiently decompose organic contaminants at varied concentrations.


 Please wait while we load your content...
Please wait while we load your content...