Persistent arsenate–iron(iii) oxyhydroxide–organic matter nanoaggregates observed in coal†
Abstract
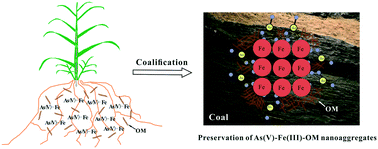
Understanding how natural nanoaggregates of iron (Fe) and organic matter (OM), currently identified in organic rich soil or peat, interact with metals and metalloids is environmentally significant. Coal is also organic-rich and exemplifies anoxic sedimentary environments with Fe usually as pyrite and not oxides. Here, we analyze the local structure of Fe (6880–21 700 mg kg−1) and As (45–5680 mg kg−1) in representative Guizhou coal samples using X-ray absorption near-edge structure and extended X-ray absorption fine structure (XANES and EXAFS) to illustrate how Fe(III) and As(V) are preserved in coal formed from reduced, organic-rich precursors. Arsenic XANES indicates that >80% of As exists as As(V) with <14% of As associated with sulfides in 5 Guizhou coal samples, confirming published but unexplained results. An As–Fe shell at 3.25–3.29 Å in the As EXAFS suggests that this As(V) is adsorbed on Fe(III) oxyhydroxides as evidenced by Fe EXAFS in these coal samples. Significantly, lower Fe–Fe coordination numbers (CN) of 0.6–1.1 relative to those in 2-line ferrihydrite (CN = 1.6) and goethite (CN = 2.1) suggest that these Fe(III) oxyhydroxides are likely Fe–OM nanoaggregates protected by OM encapsulation and adsorption of arsenate. Such structurally stabilized composites of As(V)–Fe(III)–OM may be more widely distributed and allow oxidized As and Fe to persist in other organic-rich, reducing environments.


 Please wait while we load your content...
Please wait while we load your content...