Interaction of beta-lactoglobulin and bovine serum albumin with iron oxide (α-Fe2O3) nanoparticles in the presence and absence of pre-adsorbed phosphate†
Abstract
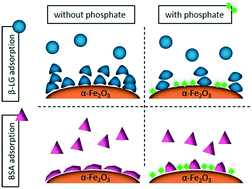
Protein adsorption onto mineral nanoparticle surfaces is critical to the function and fate of biological compounds in environmental and industrial systems. However, adsorption kinetics, coverage, and conformation of biological macromolecules are poorly understood, particularly in the presence of ubiquitous oxyanions. In this study, the adsorption of two proteins, beta-lactoglobulin (β-LG) and bovine serum albumin (BSA), onto hematite (α-Fe2O3) nanoparticles was investigated in the presence and absence of pre-adsorbed phosphate. Using solution and temporal solid-phase attenuated total reflectance-Fourier transform infrared (ATR-FTIR) spectroscopy, our results show dynamic changes in the secondary structure of both proteins when adsorbed onto nanoscale α-Fe2O3 surfaces, compared to their unbound conformations. However, these differences were attenuated in the presence of adsorbed phosphate. Adsorbed phosphate significantly reduced the protein surface coverage on iron oxide nanoparticle surfaces, and impacted protein adsorption kinetics. The latter was observed to be protein-specific, with β-LG exhibiting a higher adsorption rate and sigmoidal kinetics compared to slower, more Langmuir-type kinetics of BSA adsorption. Our results reveal the importance of phosphate on protein–mineral adsorption kinetics and conformation, a critical driver of protein function, in complex environmental systems.
- This article is part of the themed collection: Environmental Science: Nano Recent HOT Articles


 Please wait while we load your content...
Please wait while we load your content...
