Arsenic species delay structural ordering during green rust sulfate crystallization from ferrihydrite†
Abstract
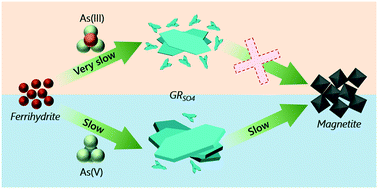
Green rust (GR) is an Fe(II)–Fe(III)-bearing phase that forms in oxygen-poor and Fe2+-rich subsurface environments where it influences trace element cycling and contaminant dynamics. GR phases have been shown to have high arsenic (As) uptake under anoxic and circum-neutral pH conditions. While geochemical controls on As uptake by GR have been identified, we still lack a fundamental understanding about GR formation in As-contaminated soils and groundwater, as well as the stability of As-bearing GR solids. In this study, we quantified the influence of As(III) and As(V) ([As]initial = 100 μM) on GR sulfate (GRSO4) crystallization during the Fe2+-induced transformation of ferrihydrite (FHY) at pH 8 (As/Fesolid = 0.008, Fe2+(aq)/Fe(III)FHY = 3). We also documented the behavior of mineral-bound As during GRSO4 crystallization and its transformation to magnetite. Our results showed that, compared to the As-free system, adsorbed As species delayed FHY transformation to GRSO4. Moreover, As(III) had a stronger inhibitory effect (at least eight-fold) than As(V) on GRSO4 crystallization, and reduced structural coherence and ordering in As(III)-bearing GRSO4 crystals. During FHY dissolution, we observed an initial release of ∼14 μM As(III) into the aqueous phase, but this was quickly adsorbed by newly-formed GRSO4 crystals. Mineral-bound As(III) resulted in at least four-fold increase in GRSO4 phase stability compared to As(V), and fully prevented its transformation to magnetite even after 720 h. Our results provide new information on the pathways of interaction of common Fe phases exposed to reducing, Fe2+-bearing and As-contaminated fluids and how these affect the structure, morphology and stability of As-bearing GR phases.


 Please wait while we load your content...
Please wait while we load your content...