Insight into bicarbonate involved efficient heterogeneous Fenton-like degradation of sulfamethoxazole over a CuFeO2 based composite under alkaline conditions†
Abstract
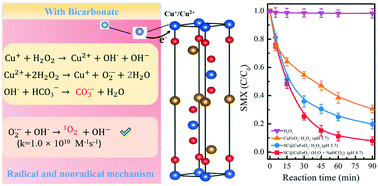
The performance of the Fenton reaction under alkaline conditions is important to promote its application in practical water treatment. In this study, a sulfide doped mesoporous carbon (SC) and CuFeO2 composite (SC@CuFeO2) was prepared and its Fenton-like activity was evaluated by the degradation of sulfamethoxazole (SMX) in an alkaline solution especially in the presence of bicarbonate. The results indicated that 93% of SMX was efficiently degraded over SC@CuFeO2 at pH 8.7 with 15 mM HCO3− in water. In comparison, the SMX removal efficiency was only about 20% without HCO3−. Furthermore, the introduction of SC can indeed improve the Fenton activity of CuFeO2. For instance, the removal efficiency of SMX over CuFeO2 and SC@CuFeO2 was 80% and 70%, respectively, under the same conditions. Based on the characterization results, electron paramagnetic resonance spectra, probe experiments and ˙OH radicals' quantitative measurement, more radical production especially the transformation processes involving reactive oxygen species (˙OH, O2˙− CO3˙− and 1O2) were responsible for the enhanced SMX degradation over SC@CuFeO2 under alkaline conditions. Finally, the intermediates of SMX degradation were detected by GC-MS/MS and the degradation pathway was discussed. These findings suggest that the effect of the water matrix on the oxidation efficiency of the Fenton reaction should be considered in real water treatment.


 Please wait while we load your content...
Please wait while we load your content...