The fate of a hazardous herbicide: a DFT-based ab initio study on glyphosate degradation†
Abstract
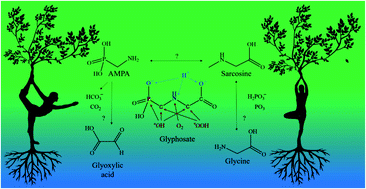
Glyphosate degradation has been extensively examined; however, only a few detailed computational studies have been performed on the topic so far. There are substantial differences between the degradation products of glyphosate, as AMPA (aminomethylphosphonic acid) is toxic while sarcosine intermediate is non-toxic. These species can have different effects on the environment and, indirectly, on the human body. We performed calculations using density functional theory and post-Hartree–Fock correlated ab initio methods to find the possible mechanisms for the degradation process by small (hydroxyl, peroxyl, and superoxide) radicals. We found that direct sarcosine formation is strongly dependent on the concentration of the radical species. AMPA and glycine were mostly formed as aldehyde derivatives, while in addition to the former, glyoxylate and bicarbonate are formed alternatively. A significant pH effect was also found for the competitive reactions determined by the calculated rate constants of the elementary steps. Overall barriers showed similarities by DFT but ab initio methods could separate them.


 Please wait while we load your content...
Please wait while we load your content...