A progressive nucleation mechanism enables stable zinc stripping–plating behavior†
Abstract
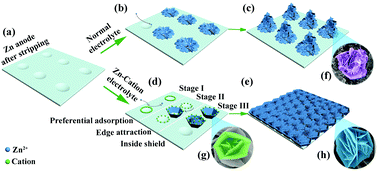
Severe zinc dendrite growth is a formidable challenge in aqueous zinc-ion batteries during repeated cycles because of the uneven distribution of electric fields on a post-stripping zinc anode. Herein, we demonstrate a totally new nucleation mechanism by introducing cations with high valence (Ce3+ and La3+) into the electrolyte to homogenize electric fields and regulate the zinc deposition behavior. Intriguingly, a detailed evolution investigation of surface morphology discloses that zinc nucleation shifts to progressive nucleation via the preferential adsorption of cation additives from instantaneous nucleation in the addtive-free electrolyte, thereby resulting in the stable zinc stripping/plating behavior. A symmetric cell with 0.01 M Ce2(SO4)3 can be cycled at 5 A g−1 with a lower voltage hysteresis of 83.3 mV for more than 700 h, which is remarkably longer than that of the pristine one (282 h). A higher capacity retention of Zn//NaV3O8·1.5H2O cell (87.0%) after 250 cycles at 5 A g−1 is also achieved in comparison with that in addtive-free electrolyte (68.1%). This work provides new and deep insights into the effects of cation adsorption on zinc nucleation.


 Please wait while we load your content...
Please wait while we load your content...