Homogenous metallic deposition regulated by defect-rich skeletons for sodium metal batteries†
Abstract
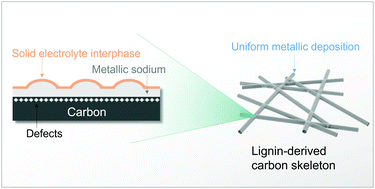
Sodium metal batteries are attracting increasing attention on account of their high energy densities as well as the abundance of sodium-based resources. However, the uneven metallic deposition and dendrite formation during cycling hinder the application of sodium metal anodes. Carbon skeletons have been reported in the literature to mitigate the dendrite formation during the plating and stripping of metallic sodium. Still, the role played by different carbon structural features (i.e., pores vs. defects) and relative mechanisms are not well understood, preventing the controllable interface engineering at the anode side. Here, we have rationally designed the structural features of sustainable carbon skeletons from a renewable precursor to unveil the roles of defects and pores for metallic deposition. The obtained carbon skeleton with rich defects and negligible pores exhibits the best performance when applied to protect metal anodes. After long cycling (>1200 hours), the retained high Coulombic efficiency (∼99.9%) of the plating and stripping processes indicates the importance of defects for inducing uniform metallic deposition. Combined with different types of cathodes (e.g., Prussian blue and sulfur), “anode-less” sodium metal batteries with enhanced electrochemical performance are also demonstrated in terms of sustainability.
- This article is part of the themed collection: Battery science and technology – powered by chemistry


 Please wait while we load your content...
Please wait while we load your content...