Understanding synergistic metal–oxide interactions of in situ exsolved metal nanoparticles on a pyrochlore oxide support for enhanced water splitting†
Abstract
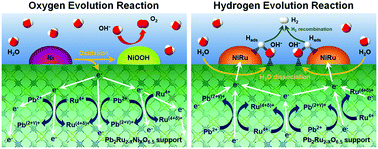
Hybrid catalysts consisting of metal nanoparticles on a metal oxide support have emerged as a new class of catalysts that can improve various electrocatalytic reactions, but the origin of the improved performance is still unclear. Here we demonstrate that rationally designed hybrid catalysts through the in situ exsolution process of metallic nanoparticles on a B-site Ni-substituted lead ruthenate pyrochlore oxide improve both oxygen evolution reaction and hydrogen evolution reaction activity. A combination of operando X-ray absorption spectroscopy measurements and density functional theory calculations reveals that the generated oxygen and cation vacancies in the pyrochlore oxide support during the in situ exsolution process can decrease the charge-transfer energy, thereby facilitating charge transfer between exsolved metal nanoparticles–oxide support as well as hybrid catalyst–electrolyte. These findings establish a structure–property relationship of complicated hybrid catalysts for efficient water splitting, suggesting a new strategy in designing various hybrid catalysts for other electrochemical reactions.


 Please wait while we load your content...
Please wait while we load your content...