Lithium-activated SnS–graphene alternating nanolayers enable dendrite-free cycling of thin sodium metal anodes in carbonate electrolyte†
Abstract
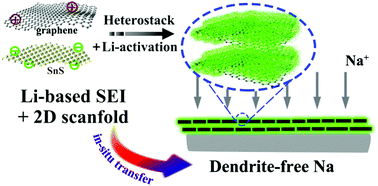
Sodium metal battery (SMB, NMB) anodes can become dendritic due to an electrochemically unstable native Na-based solid electrolyte interphase (SEI). Herein Li-ion activated tin sulfide graphene nanocomposite membrane (A-SnS–G) is employed as an artificial SEI layer, allowing cyclability of record-thin 100 μm Na metal foils. The thin Na metal is prepared by a self-designed metallurgical rolling protocol. A-SnS–G is initially placed onto the polypropylene (PP) separator but becomes in situ transferred onto the Na metal surface. Symmetric metal cells protected by A-SnS–G achieve low-overpotential extended high-rate cycling in a standard carbonate electrolyte (EC : DEC = 1 : 1, 5% FEC). Accumulated capacity of 1000 mA h cm−2 is obtained after 500 cycles at 4 mA cm−2, with accumulated capacity-to-foil capacity (A/F) ratio of 90.9. This is among the most favorable cycle life, accumulated capacity, and anode utilization combinations reported. Protection by non-activated SnS–G membrane yields significantly worse cycling, albeit still superior to the baseline unprotected sodium. Post-mortem and dedicated light optical analysis indicate that metal swelling, dendrite growth and dead metal formation is extensive for the unprotected sample, but is suppressed with A-SnS–G. Per XPS, post-100 cycles near-surface structure of A-SnS–G is rich in metallic Sn alloys and inorganic carbonate salts. Even after 300 cycles, Li-based SEI components ROCO2-Li, Li2CO3 and LiF are detected with A-SnS–G. As a proof of principle, an SMB with a high mass loading (6 mg cm−2) NVP cathode and a A-SnS–G protected anode delivered extended cyclability, achieving 74 mA h g−1 after 400 cycles at 0.4C.


 Please wait while we load your content...
Please wait while we load your content...