Measurements of the partitioning of nitric acid and sulfuric acid in aqueous/organic phase-separated systems†
Abstract
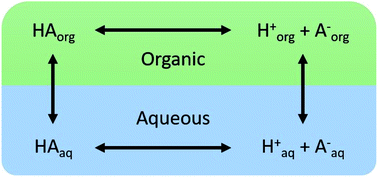
Partitioning of nitric acid and sulfuric acid between aqueous and organic phases may play a role in the chemistry of aqueous/organic phase-separated atmospheric aerosol particles, as well as other environmental systems. To assess the extent to which these acids partition to organic phases, organic compounds with O : C ratios ranging from 0 to 0.75 were mixed with acidified aqueous phases. The organic phase was removed and its acid content quantified with a mass-based measurement. Up to 60% of the acid was observed to partition to the organic phase, present either entirely in the dissociated form, the undissociated form, or both depending on the organic phase. Values of partitioning coefficients, Ddiss and Dun, corresponding to partitioning of the dissociated and undissociated acid, were determined by fitting a simple partitioning model to the data. Values of Dun correlated well with the O : C ratio of the organic component and to a lesser extent with the water content of the organic phase. The addition of ammonium sulfate led to an increase or decrease in partitioning of sulfuric acid to the organic phase, apparently depending on the balance between the resulting enhancement in the HSO4− concentration and the reduction in the water content of the organic phase. These results demonstrate that significant fractions of sulfuric and nitric acid can partition to the organic phase in phase-separated systems in undissociated and dissociated forms, and thus potentially participate in specific or general acid catalysis of organic reactions.


 Please wait while we load your content...
Please wait while we load your content...