Hypercoordinated eka-tin materials with dangling aryl-methoxy and -methylthio ligands exhibiting intramolecular secondary bonding and aryl bond stabilization in reactions with organotin chlorides†
Abstract
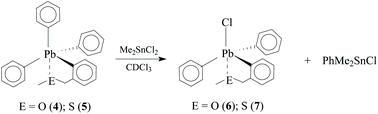
The syntheses of [2-(CH3ECH2)C6H4]PbPh3−nCln, (n = 0, E = O (4), E = S (5); n = 1, E = O (6), E = S (7); n = 2, E = O (8), are described. NMR and single crystal data illustrate significant Pb⋯E interactions increasing as n progresses from 0 to 2. The Pb⋯E interactions stabilize the Pb–aryl bonding to the extent that the reactions of 4 and 5 with Me2SnCl2 result in interchange of a Ph group and Cl to produce 6 and 7, respectively, together with Me2PhSnCl.
- This article is part of the themed collection: Tin: Modern chemistry of an element from antiquity


 Please wait while we load your content...
Please wait while we load your content...