Cell-permeable lanthanide–platinum(iv) anti-cancer prodrugs†
Abstract
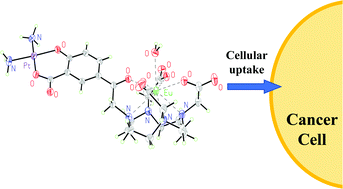
Platinum compounds are a vital part of our anti-cancer arsenal, and determining the location and speciation of platinum compounds is crucial. We have synthesised a lanthanide complex bearing a salicylic group (Ln = Gd, Eu) which demonstrates excellent cellular accumulation and minimal cytotoxicity. Derivatisation enabled access to bimetallic lanthanide–platinum(II) and lanthanide–platinum(IV) complexes. Luminescence from the europium–platinum(IV) system was quenched, and reduction to platinum(II) with ascorbic acid resulted in a “switch-on” luminescence enhancement. We used diffusion-based 1H NMR spectroscopic methods to quantify cellular accumulation. The gadolinium–platinum(II) and gadolinium–platinum(IV) complexes demonstrated appreciable cytotoxicity. A longer delay following incubation before cytotoxicity was observed for the gadolinium–platinum(IV) compared to the gadolinium–platinum(II) complex. Functionalisation with octanoate ligands resulted in enhanced cellular accumulation and an even greater latency in cytotoxicity.


 Please wait while we load your content...
Please wait while we load your content...