Ternary alkali ion thiogallates, A5GaS4 (A = Li and Na), with isolated tetrahedral building units and their ionic conductivities†
Abstract
Two new ternary thiogallates in the A5GaS4 (A = Li (I) and Na (II)) series have been synthesized for the first time employing a gas passing route using oxide precursors and a high temperature solid state route using stoichiometric combinations of elements, respectively. Li5GaS4 crystallizes in the P21/m space group and the structure is built up of layers of corner sharing tetrahedra of LiS4 and GaS4 stacked along the a-axis and the octahedrally coordinated Li ions residing in the interlayer space. Na5GaS4 crystallizes in the Pbca space group and the structure consists of isolated (GaS4)5− tetrahedra held together by charge balancing sodium ions in distorted tetrahedral and octahedral coordination geometries. Measurements of ionic conductivity of the compounds showed room temperature ionic conductivities of 1.8 × 10−7 and 4.0 × 10−7 S cm−1 with activation energies of 0.54 and 0.28 eV, respectively, for I and II. Density functional theory calculations show close agreement in structural parameters with the measured data and predict band gaps of 2.75 eV (I) and 2.70 eV (II). Single point hybrid functional calculations result in band gaps of 3.95 and 3.65 eV correspondingly, in better agreement with the experimental value of ∼4.1 eV for both. Bond valence energy landscape maps suggest the absence of any suitable diffusion path for Li in Li5GaS4. On the other hand, BVEL maps of Na5GaS4 confirm that the tetrahedrally coordinated Na ions are responsible for ionic conduction, whereas the involvement of octahedrally coordinated Na ions in the conduction process could not be discerned.
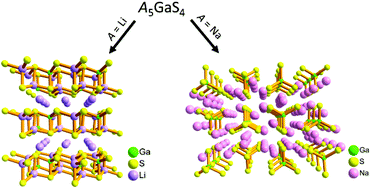


 Please wait while we load your content...
Please wait while we load your content...