Cu oxide deposited on shape-controlled ceria nanocrystals for CO oxidation: influence of interface-driven oxidation states on catalytic activity†
Abstract
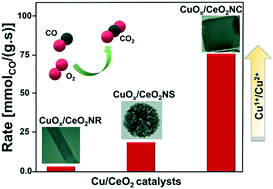
The design of a catalyst with a highly active and stable oxidation state is of great interest in heterogeneous catalysis. Herein, the relationship between catalytic activity and oxidation state on Cu deposited on CeO2 nanocrystals has been elucidated by varying the shape of the ceria (CeO2) support. Three types of CeO2 nanocrystals were prepared for supporting Cu oxide (CuOx): CeO2 nanocubes (NCs), nanorods (NRs) and nanospheres (NSs). The Cu oxide deposited on CeO2NC has shown higher CO oxidation activity at a lower temperature than that over the NR and NS surfaces. Furthermore, characterization of structure and oxidation states revealed that the stable Cu1+ oxidation state on the surface of CuOx/CeO2NC formed at a low loading of copper (∼1.5 wt%), which acts as an active site for the CO oxidation. In contrast to the high surface area and redox properties, a systematic catalytic activity trend was observed among the catalysts with the extent of the Cu1+ oxidation state. We demonstrate that the polar (100) surface facets of NCs contribute significantly to the formation of surface hydroxyl groups, which are required for the selective and stable Cu1+ state at a low loading.


 Please wait while we load your content...
Please wait while we load your content...