Fabrication of Pd/CeO2 nanocubes as highly efficient catalysts for degradation of formaldehyde at room temperature†
Abstract
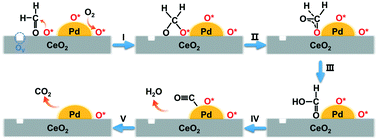
Eliminating indoor formaldehyde (HCHO) pollution at room temperature is highly needed to improve the environment while saving energy. Herein, Pd/CeO2 catalysts with different morphologies of the CeO2 support were employed to investigate their performance for HCHO oxidation. The Pd/CeO2 nanocube (Pd/CeO2-C) catalyst exhibited excellent catalytic activity compared to commercial CeO2 and CeO2 nanorods. HCHO can be completely converted into CO2 and H2O over the Pd/CeO2-C (0.64 wt% Pd) catalyst at a weight hourly space velocity of 36 000 mL g−1 h−1 at room temperature. Multiple characterization results confirmed that the strong interfacial interaction between Pd and the CeO2-C support guarantees a high percentage of metallic Pd on the CeO2-C surface and its high dispersion, which is crucial for the oxidation of formaldehyde at low temperature. The in situ diffuse reflectance infrared Fourier transform results indicated that CO, formate and dioxymethylene species are the main intermediates over the Pd/CeO2-C catalyst, and the active O species is responsible for their decomposition and oxidation. This work may promote the practical application of high performance Pd-based catalysts for indoor HCHO purification and related catalytic processes.


 Please wait while we load your content...
Please wait while we load your content...